Prof. Dr. Delia Brauer
Image: PrivateProf. Dr. Delia BRAUER
Email: delia.brauer@uni-jena.de
Phone: +49 3641-9-48510
Delia Brauer is Professor of Bioactive Glasses. She is a Fellow of the Society of Glass Technology and member of its Board of Fellows. She chairs the Technical Committee 04 (Glasses as Biomaterials) of the International Commission on Glass (ICG) and is a member of the Basic Sciences and Technology Committee of the Society of Glass Technology. She is also a member of the Jena Center for Microbial Communication (JCMC).
Over the last years, rare earth-doped fluoride crystals within an aluminosilicate glass matrix have gained much interest, as they combine the benefits of fluoride crystals (e.g. low phonon energy) with the thermal and chemical stability of aluminosilicate glasses. Most of these glasses, however, have very high melting temperatures (around 1600°C), which is a disadvantage from a technological viewpoint. One current research project therefore focuses on designing rare earth-doped fluoro-aluminosilicate glasses of lower melting temperatures and characterising their crystallisation behaviour.
Compared to silicate glasses, phosphate glasses have relatively high refractive indices and low dispersion. One disadvantage, however, is their poor durability in the presence of water and humidity. While it is known that the solubility of phosphate glasses can vary over several orders of magnitude, there are still many open questions as to their dissolution mechanism. We therefore currently investigate the details of phosphate glass dissolution, as an improved understanding of phosphate glass dissolution can pave the way towards more chemically stable compositions.
Research Areas
Our research focuses on the materials chemistry of inorganic, non-metallic materials, especially glasses and glass-ceramics, with a particular focus on the interaction between materials and water. She is also interested in how glass composition, structure and properties are connected, particularly in glasses having a highly disrupted structure. Current research areas include:
- Improving the hydrolytic stability of phosphate glasses
- Glass-based cement systems
- Bioactive glasses with improved processing
- Morphology and topography of glass-ceramic surfaces
- Synchrotron applications for glasses and glass-ceramics
- Characterisation of archaeological glasses, glazes and ceramics
- 3D-μCT in bioactive glass research
Teaching Fields
Prof. Brauer teaches students of the courses Materials Science, Chemistry and Chemistry of Materials at the Bachelor and Master level. Her courses include:
- General and inorganic chemistry
- Materials science (courses on glass, ceramics, glass structure and ceramics in medicine)
- Materials synthesis and characterization
Research Methods
Prof. Brauer‘s research group prepares and characterizes glasses and glass-ceramics. Techniques include:
- Equipment for glass melting
- High-temperature characterization including DSC, heating microscopy
- Structural characterisation including x-ray diffraction, FTIR and Solid-State (MAS) NMR spectroscopy, 3D x-ray microscopy
- Equipment for dissolution experiments and analysis
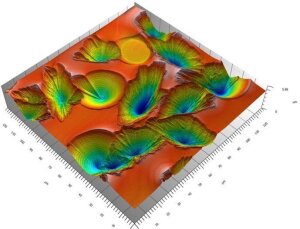
Figure: LSM picture of fresnoite crystals on a glass surface with the crystals sinking into the bulk (blue is deep, red is the surface).
Picture: Brauer research group.Recent Research Results
Bioactive Glasses
Bioactive silicate glasses are used clinically to regenerate bone and as dentifrices to re-mineralize teeth. Prof. Brauer’s group investigates how glass structure controls ion release, dissolution and crystallisation; and how a structure-based glass design allows for optimisation of properties.
Structure and mechanical properties of aluminosilicate glasses
Aluminosilicate glasses are used for various applications where mechanical performance is key, e.g. as for mobile phone displays for fibres in glass fibre composites. We have shown that incorporation of phosphate, as an additional network former, offers unique opportunities for finetuning glass properties, e.g. elastic properties and hardness, via changes in glass structure and polymerization [2].
Surface crystallisation and topography
As glasses are thermodynamically not stable, heat-treatment causes crystallization. Using a barium titanium silicate system as an example, we study the influence of atmosphere composition (moisture, air, argon or vacuum) on surface crystallisation of fresnoite crystals.
[1] Wetzel et al., Sci. Rep. 10, 15964 (2020).
[2] Grammes et al., Front. Mater. 7, 115 (2020).